Abstract
Background: Although there have been significant recent advances in the treatment of relapsed/refractory (r/r) large B-cell lymphoma (LBCL), there remains a substantial unmet need to optimize the frontline management of high-risk LBCL. Patients with high International Prognostic Index (IPI) scores 4-5 have particularly poor progression-free survival (PFS) rates of ~50% with R-CHOP, and 2-year PFS may be as low as 10-25% for patients with positive interim PET scans. In the phase III DLCL04 and SWOG-9704 trials, the use of upfront consolidative autologous stem cell transplantation (ASCT) after R-CHOP resulted in significant improvements in PFS, with the latter study also demonstrating a significant improvement in overall survival (OS) in the age-adjusted high IPI subgroup. Furthermore, the GAINED trial demonstrated favorable outcomes using an interim PET-adapted strategy, where consolidative ASCT appeared to overcome the poor prognostic impact of a positive interim PET. However, there is only limited and conflicting data from prospective or retrospective studies concerning ASCT consolidation for the highest risk patients with LBCL and IPI score 4-5, particularly those with positive interim PET scans.
Methods: This retrospective, multicenter study included consecutive patients with newly diagnosed LBCL and high IPI score 4-5 who received consolidative ASCT as a planned part of first-line therapy at the Tom Baker Cancer Centre and Cross Cancer Institute in Alberta, Canada. Patients were excluded if they had primary central nervous system (CNS) lymphoma, previously treated indolent B-cell lymphoma, or underwent ASCT for r/r LBCL. PFS, OS, and disease-specific survival (DSS) were determined using the Kaplan-Meier method.
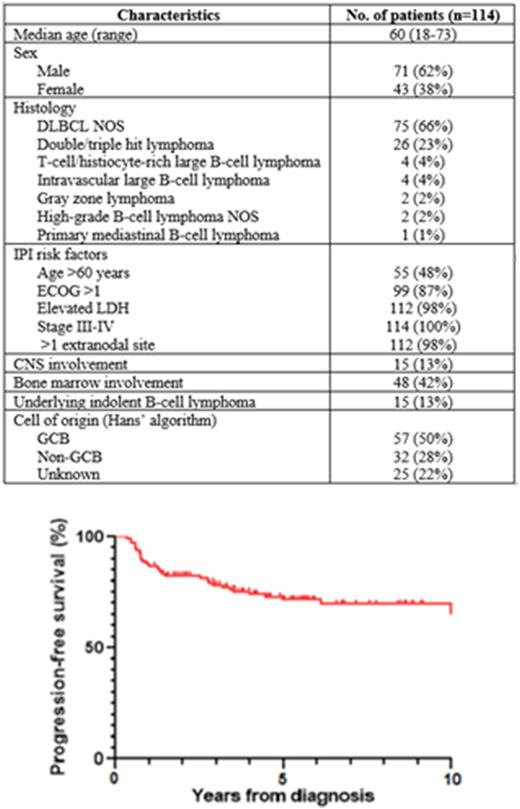
Results: We included 114 patients with median age 60 years (range 18-73) and median follow-up time 5.6 years. High-risk features at diagnosis included IPI score 4 in 81 (71%), IPI score 5 in 33 (29%), stage IV in 112 (98%), >1 extranodal site in 109 (96%), non-GCB cell of origin by Hans’ algorithm in 32 (28%), double-hit lymphoma in 26 (23%), and secondary CNS involvement in 15 (13%). The most common induction regimen was R-CHOP (n=102) and 25 (22%) patients received 1-3 cycles, 71 (62%) received 4 cycles, and 18 (16%) received 5-6 cycles of induction. Rituximab with dose-intensive cyclophosphamide, etoposide, and cisplatin (R-DICEP) was frequently used (n=69) to intensify induction and for peripheral blood stem cell mobilization. Interim PET was performed during induction for 45 patients and was positive (Deauville 4-5) in 24 (53%) and negative (Deauville 1-3) in 21 (47%). Pre-ASCT disease status was complete response (CR) in 68 (60%), partial response (PR) in 43 (38%), and unknown in 3 (3%) patients. The most common ASCT conditioning regimens were BEAM in 41 (36%), BuMel in 39 (34%), and TBM in 13 (11%) patients.
At 2 years, PFS was 82% (95% CI 74-88%), OS was 86% (95% CI 78-91%), and DSS was 87% (95% CI 79-92%). At 5 years, PFS was 72% (95% CI 62-80%), OS was 74% (95% CI 64-81%), and DSS was 80% (95% CI 71-87%). Relapse occurred in 21 (18%) patients at median 284 days (range 168-2229) after ASCT. There was 1 (1%) death due to early transplant-related infection and 3 (3%) late deaths from pulmonary fibrosis and therapy-related myeloid neoplasms. There were no significant differences in DSS for patients with versus without CNS involvement (77% vs. 80%, p=0.62), double-hit lymphoma versus DLBCL NOS (85% vs. 78%, p=0.80), GCB versus non-GCB cell of origin (78% vs. 80%, p=0.89) and positive versus negative interim PET (83% vs. 74%, p=0.51).
Discussion: In this study of 114 selected patients with high-risk LBCL with IPI score 4-5, consolidative ASCT was associated with excellent outcomes with long-term disease-specific survival in 80%. Importantly, the efficacy of consolidative ASCT appeared to be independent of adverse prognostic factors including CNS involvement, double-hit status, non-GCB cell of origin, and interim PET positivity. The results of our study are consistent with those of previous clinical trials and provide real-world evidence confirming that consolidative ASCT can be considered a reasonable treatment option for eligible patients with high-risk LBCL.
Disclosures
Puckrin:Beigne: Honoraria. Sterrett:Abbvie: Honoraria. Chua:Pfizer: Honoraria; Bayer: Honoraria; Gilead: Honoraria; Merck: Honoraria; Amgen: Honoraria; Incyte: Honoraria; Abbvie: Honoraria; Seagen: Honoraria. Owen:Incyte: Honoraria; GIlead: Honoraria; Roche: Honoraria; Merck: Honoraria; AstraZeneca: Honoraria; Janssen: Honoraria; Novartis: Honoraria; AbbVie: Honoraria; BeiGene: Honoraria. Shafey:BeiGene: Honoraria; Janssen: Honoraria; Astrazeneca: Honoraria; Novartis: Honoraria; BMS: Honoraria; Kite/Gilead: Honoraria; Incyte: Honoraria. Stewart:Gilead: Honoraria; Novartis: Honoraria; Beigene: Honoraria; Janssen: Honoraria; Roche: Honoraria; Seagen: Honoraria; Apobiologix: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal